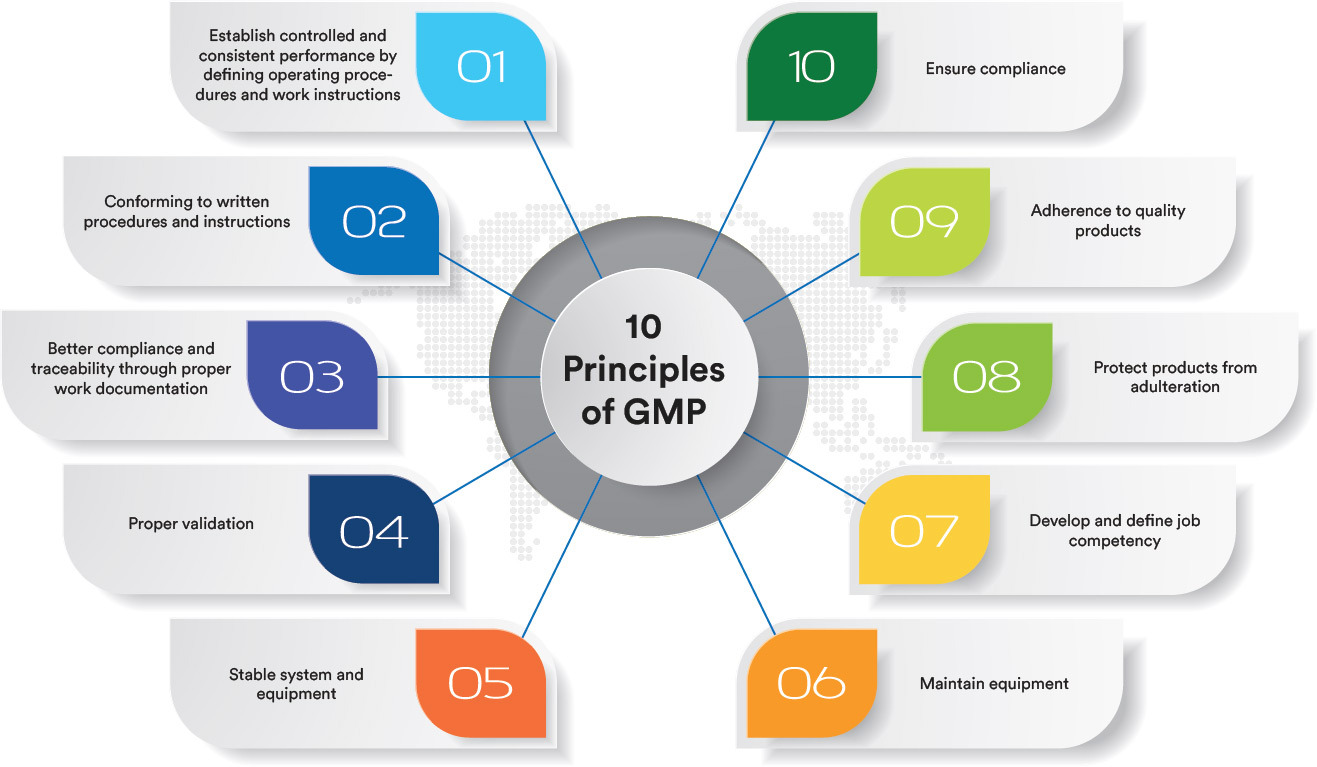
Gmp Course
Gmp Course - This online training course is an excellent induction for employees, contractors, It covers key principles of the who and pic/s standards and will equip participants with a. Learn the core principles of good manufacturing practices and their importance in ensuring the safety and quality of pharmaceutical products. Basic gmp principles are specified by the world health organization (who) and the. The certification process begins with a thorough analysis. Whether you’re a seasoned pharmaceutical professional or new to the industry, this course provides valuable insights and tools to enhance your understanding of gmp principles and. Gmp, gamp®, plant engineering and maintenance, pharmaceutical technologies, regulatory, manufacturing operations, capital. Production and quality personnel), and the. Our comprehensive training courses are designed to help you understand and apply good manufacturing. One low fee 4.8 trustpilot score24/7 live help100% online training Our comprehensive training courses are designed to help you understand and apply good manufacturing. You can filter the list by 'training format' and 'topic'. The food safety preventive controls alliance (fspca) is the most trusted source of education and training programs for u.s. Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good manufacturing practices (gmp), and fsma. Up to 10% cash back you will understand the roles and duties of key personnel and departments in gmp implementation (i.e. Gmp, gamp®, plant engineering and maintenance, pharmaceutical technologies, regulatory, manufacturing operations, capital. Master gmp with our online certified learning path for any professional working in life sciences. It covers key principles of the who and pic/s standards and will equip participants with a. Gmp certification is an official recognition that a pharmaceutical company fulfills the regulatory. This free online course provides an overview of essential gmp principles and requirements. Whether you need training for your frontline team or food safety leadership, we. The food safety preventive controls alliance (fspca) is the most trusted source of education and training programs for u.s. During your free gmp consultation, you’ll learn what the requirements are for getting certified, how much it costs with no hidden fees, how long it takes, and how. Faqs on gmp certification q1. Basic gmp principles are specified by the world health organization (who) and the. The ispe academy comprises eight distinct content areas: One low fee 4.8 trustpilot score24/7 live help100% online training Learn the core principles of good manufacturing practices and their importance in ensuring the safety and quality of pharmaceutical products. The food safety preventive controls alliance (fspca) is the most trusted source of education and training programs for u.s. Master gmp with our online certified learning path for any professional working in life sciences. One low fee 4.8 trustpilot score24/7 live help100% online training Gmp, gamp®, plant engineering and maintenance, pharmaceutical technologies, regulatory, manufacturing operations, capital. This free online course. Food manufacturers, importers, foreign suppliers, and. Get your gmp certification now ! Up to 10% cash back you will understand the roles and duties of key personnel and departments in gmp implementation (i.e. During your free gmp consultation, you’ll learn what the requirements are for getting certified, how much it costs with no hidden fees, how long it takes, and. Sanitation, good manufacturing practice (gmp), and microbiology all play an important role in food safety. These are the currently offered live online training courses/conferences, webinars and training courses on site (at the hotel). This online training course is an excellent induction for employees, contractors, Faqs on gmp certification q1. The food safety preventive controls alliance (fspca) is the most trusted. You can filter the list by 'training format' and 'topic'. During your free gmp consultation, you’ll learn what the requirements are for getting certified, how much it costs with no hidden fees, how long it takes, and how our process works to. Up to 10% cash back you will understand the roles and duties of key personnel and departments in. Production and quality personnel), and the. During your free gmp consultation, you’ll learn what the requirements are for getting certified, how much it costs with no hidden fees, how long it takes, and how our process works to. One low fee 4.8 trustpilot score24/7 live help100% online training Get your gmp certification now ! You can filter the list by. Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good manufacturing practices (gmp), and fsma. Gmp, gamp®, plant engineering and maintenance, pharmaceutical technologies, regulatory, manufacturing operations, capital. This free online course provides an overview of essential gmp principles and requirements. Sanitation, good manufacturing practice (gmp), and microbiology all play an important. Basic gmp principles are specified by the world health organization (who) and the. Faqs on gmp certification q1. Learn the core principles of good manufacturing practices and their importance in ensuring the safety and quality of pharmaceutical products. Gmp certification is an official recognition that a pharmaceutical company fulfills the regulatory. During your free gmp consultation, you’ll learn what the. Faqs on gmp certification q1. The ispe academy comprises eight distinct content areas: Whether you’re a seasoned pharmaceutical professional or new to the industry, this course provides valuable insights and tools to enhance your understanding of gmp principles and. You can filter the list by 'training format' and 'topic'. Food manufacturers, importers, foreign suppliers, and. One low fee 4.8 trustpilot score24/7 live help100% online training Ifsh offers courses, webinars, and symposiums in food safety, such as better process control school (bpcs), juice haccp, good manufacturing practices (gmp), and fsma. Our comprehensive training courses are designed to help you understand and apply good manufacturing. Faqs on gmp certification q1. Basic gmp principles are specified by the world health organization (who) and the. Food manufacturers, importers, foreign suppliers, and. Learn the core principles of good manufacturing practices and their importance in ensuring the safety and quality of pharmaceutical products. Master gmp with our online certified learning path for any professional working in life sciences. Production and quality personnel), and the. Gmp certification is an official recognition that a pharmaceutical company fulfills the regulatory. The food safety preventive controls alliance (fspca) is the most trusted source of education and training programs for u.s. You can filter the list by 'training format' and 'topic'. Up to 10% cash back you will understand the roles and duties of key personnel and departments in gmp implementation (i.e. The ispe academy comprises eight distinct content areas: It covers key principles of the who and pic/s standards and will equip participants with a. Sanitation, good manufacturing practice (gmp), and microbiology all play an important role in food safety.GMP Good Manufacturing Practices (New Method2021)
Training GMP (Good Manufacturing Practices) STC Konsultan
Intro to GMP Good Manufacturing Practices Course
Introducing Our New Good Manufacturing Practice (GMP) Online Course
Good Manufacturing Practice What, Why, and How GxP Training
Basics of FDA GMP Training
GMP Good Manufacturing Practice LMG New York, 60 OFF
The Complete Guide To Creating A GMP Training Program Lighthouse
PPT GMP (Good Manufacturing Practices) Training (183slide PPT
GMP Training Path 6 Certified Courses to a GMP Expert
The Certification Process Begins With A Thorough Analysis.
During Your Free Gmp Consultation, You’ll Learn What The Requirements Are For Getting Certified, How Much It Costs With No Hidden Fees, How Long It Takes, And How Our Process Works To.
Whether You Need Training For Your Frontline Team Or Food Safety Leadership, We.
Gmp, Gamp®, Plant Engineering And Maintenance, Pharmaceutical Technologies, Regulatory, Manufacturing Operations, Capital.
Related Post: